Approach to Therapy
Alzheimer’s Disease & The Metabolic Approach to Therapy
Effective disease-modifying therapy is critically needed for the treatment of Alzheimer’s disease (AD), as the prevailing thinking over the last two decades has thus far not resulted in breakthrough solutions. Despite extensive research and treatment with drugs that target symptoms, including acetylcholinesterase inhibitors and NMDA receptor antagonists the clinical course of Alzheimer’s disease has not been meaningfully slowed or stopped.
Inherent in AD are 2 major metabolic dysfunctions: Aberrant glucose energy & lipid metabolism that lead to the pathological sequelae of plaques, tangles, fat deposits, oxidative stress and inflammation. These pathologies in turn perpetuate glucose energy and lipid metabolism dysfunctions in a massive positive feedback loop that lead to brain atrophy and death.
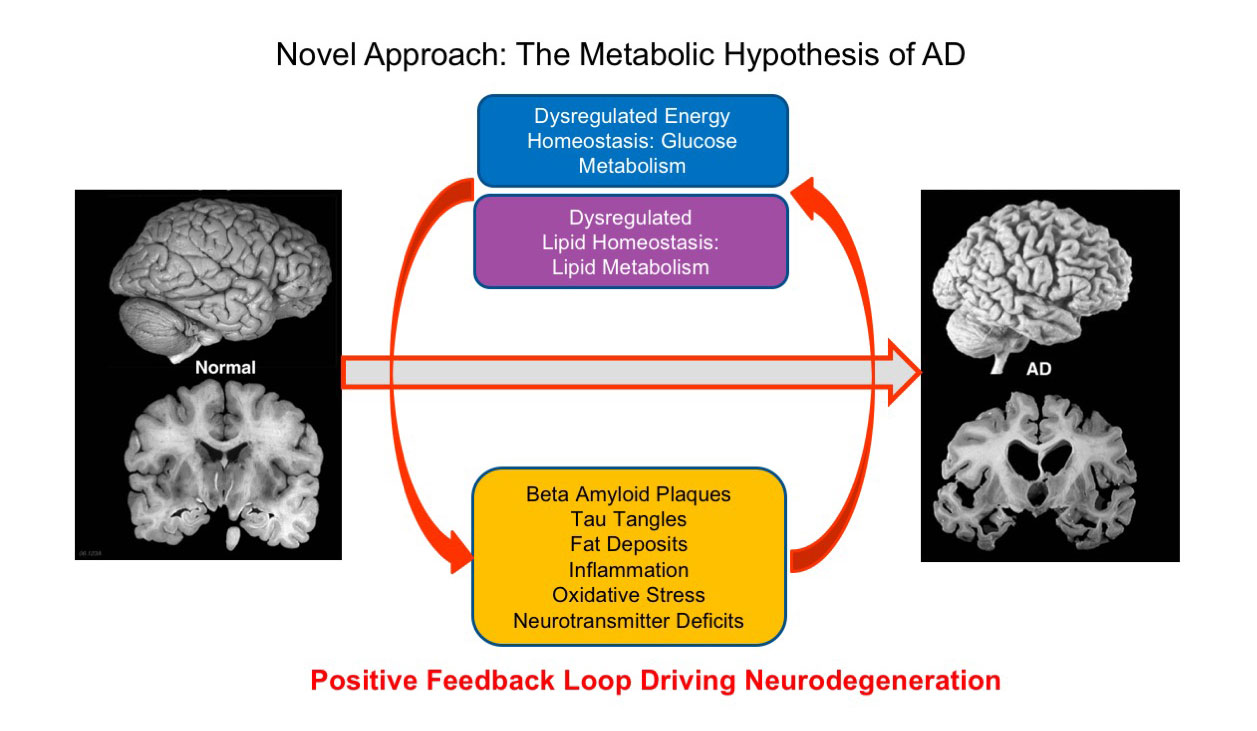
The rationale behind T3D-959 is to break this cycle by correcting these metabolic defects to effect potential disease remediation.
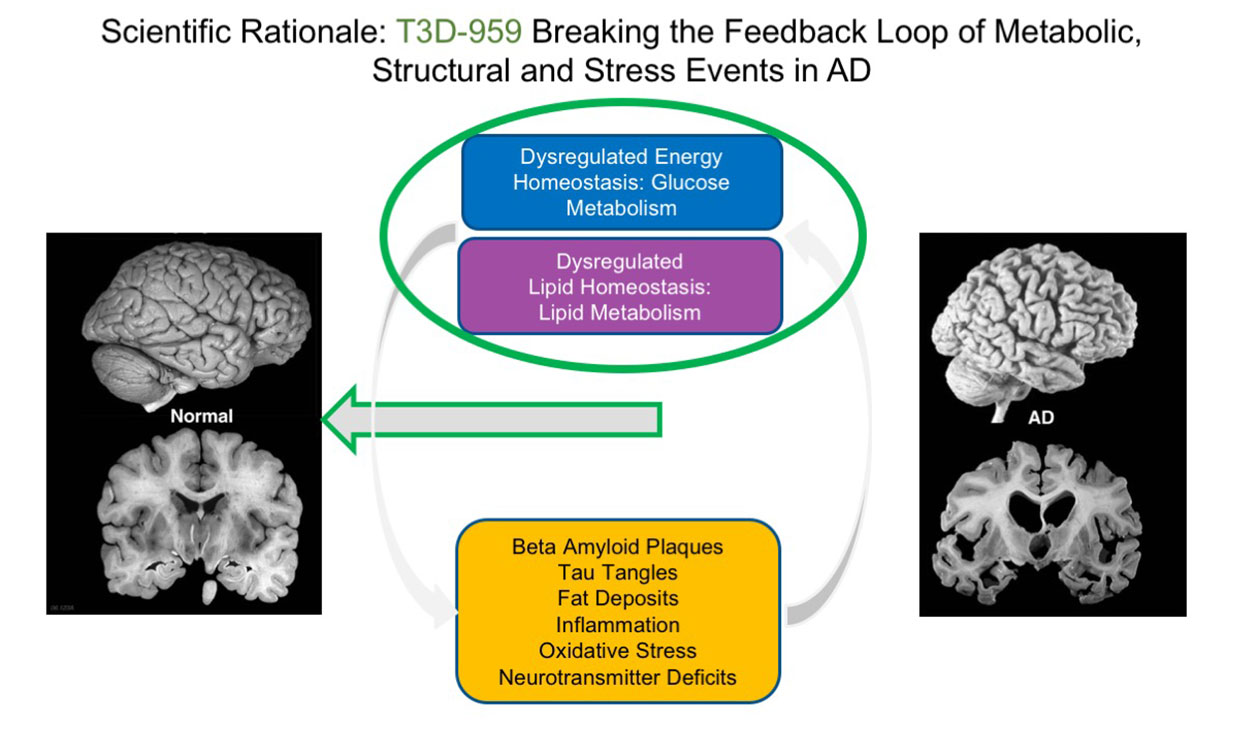
In addition to the aforementioned feedback loop, the Company’s novel neurometabolic approach to treating AD recognizes:
- Metabolism alterations precede structural change in AD (See FDG-PET image below).
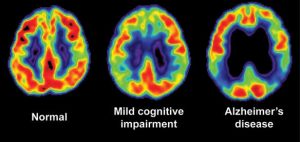
- Impaired glucose metabolism is inherent in neurodegeneration, found not just in AD, but in Huntington’s Disease and Parkinson’s Disease as well.
- Aberrant lipid metabolism was recognized by Alois Alzheimer 100y ago, observing fat deposits in the brain, identified last year as triglycerides. Also, the lipid trafficking ApoE as a factor in AD is well established.
- Abeta and Insulin have interwined molecular interactions with Abeta binding to insulin, the insulin receptor, insulin degrading enzyme and regulating IRS-1.
- Similarities in AD and Type 2 diabetes that both involve insulin resistance; including amyloid aggregation and cognitive impairment.
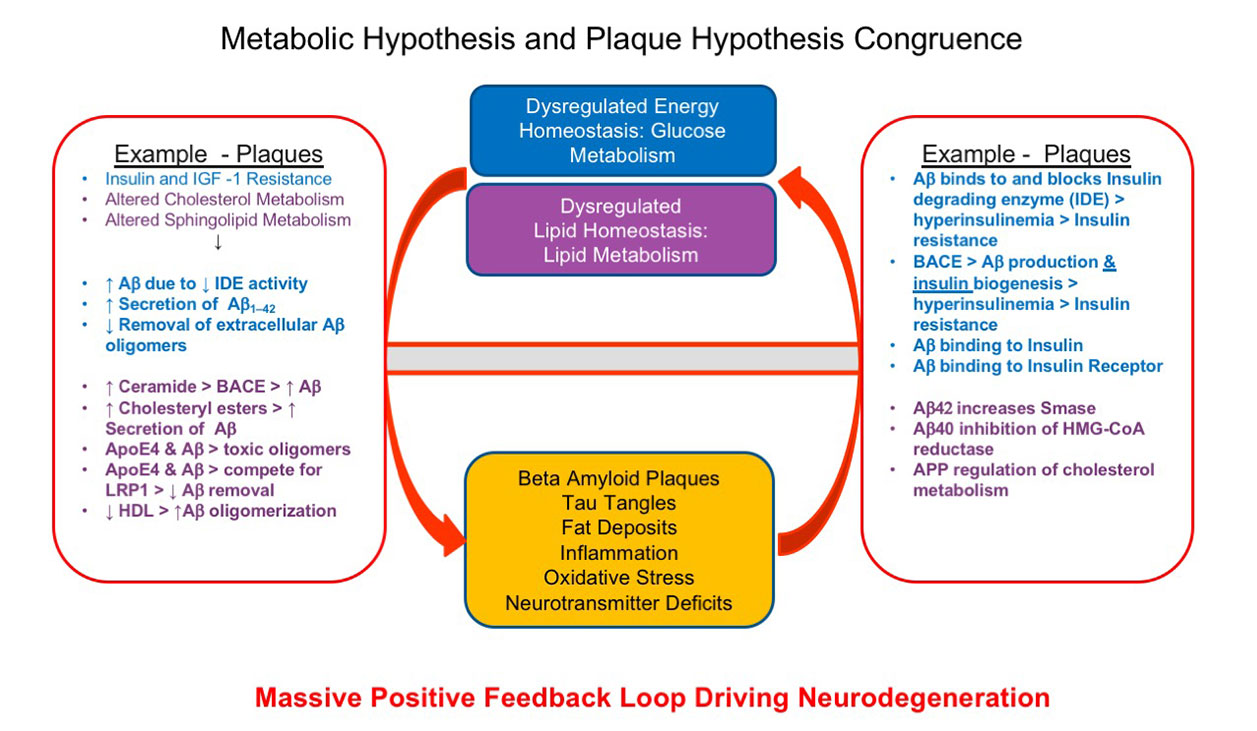
Congruence of The Metabolic Approach and Plaque Approach to AD Therapy
The metabolic hypothesis and beta amyloid plaque hypothesis of AD are not antithetical but congruent. Approaching disease therapy directed at ameliorating both metabolic dysfunctions and plaque accumulation is a potentially attractive avenue to effect potential disease remediation.

Alzheimer's disease
In pre-clinical animal studies, T3D Therapeutics’ new drug candidate, T3D-959, has shown evidence of a reversal of the diseased state in the brain by histopathology (microscopic tissue analysis).

Mild Cognitive Impairment

Other Neurodegenerative Diseases
PPAR delta agonist compounds such as T3D-959 have recently been found to have strong neuroprotective effects that we believe make T3D-959 a candidate drug for development for potential treatment in a number of CNS diseases (e.g. Huntington’s disease, Parkinson’s disease) (refs.15,16).
Beyond Alzheimer’s disease, these other diseases present added life cycle management opportunities for T3D-959 and other structurally-related compounds within the Company’s exclusive license.
